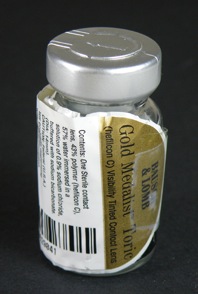
Bausch & Lomb Toric Gold Medalist Vial
Catalogue Number: 2454 Bausch & Lomb Toric Gold Medalist Vial Category: Spectacles and lenses Sub-Category: Contact lenses and accessories Corporation: Bausch & Lomb Incorporated, Rochester, New York. Year Of Publication/Manufacture: 1988 to 1992 Time Period: 1940 to 1999 Place Of Publication/Manufacture: Rochester, New York, USA Publisher/Manufacturer: Bausch & Lomb Incorporated Description Of Item: One vial capped with silver top, with a label taped on which contains the words: Bausch & Lomb Gold Medalist Toric (hefilcon C) Visibility Tinted Contact Lens. Contents: One Sterile contact lens 43% polymer (hefilcon C) 57% water immersed in a solution of 0.9% sodium chloride buffered with sodium bicarbonate. (Dia:14 mm) CAUTION: Federal (U.S.A.) law prohibits dispensing without a prescription. Lens parameters Lot No. and Expiry date of 08/200 are included on the label. The original label on the vial, covered by the label described is that of a B & L Optima Toric (hefilcon B) 45% water. Historical Significance: Bausch & Lomb Gold Medalist Toric lenses were introduced in the period (1988 to 1996) when Bausch & Lomb was one of ten world-wide sponsors of the Summer and Winter Olympic Games as a daily wear monthly disposable lens. Trial lenses were produced in two base curves (8.30 and 8.60) 0.00 cyl and axis of 000. Orientation of the trial lens was used to ascertain the closest axis specification to the patient's prescription. Blister packs were introduced later and a change in material to Lotrafilcn A was introduced in 1992. The lenses were renamed Soflens 66 in 1997 when Olympic sponsorship ceased(See Cat No. 2453) This vial has the label from one of the blisters attached over the original label suggesting the lens has been re-packaged after trial on a patient's eye How Acquired: Donated by Karingal Optical Condition: Good, label wrapped around vial and secured with adhesive tape Location: Documents Room Small Red Cabinet Drawer 1 |